NH 4 PO 4. Which one of the following elements can form ions with more than one.
Therefore the name of HClO 3 is.
. Electron Geometry Molecular Shape d. The formula for the compound formed from the polyatomic ions NH 4 and PO 4 3 is. The carbonate ion has the formula CO32-.
The anion ClO3- is the chlorate. The chemical name for ClO 3- is chlorate ion. Here is a site that will show you how to determine the oxidation.
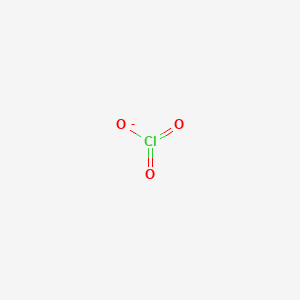
The substance ClO3-1 is best described as. Draw a Lewis structure for the chlorate ion ClO 3-in which all atoms satisfy but do not exceed the octet rule. The gain of electrons is reduction.
According to the VSEPR theory the geometry of the SO 3 molecule is A pyramidal. All other atoms do not have charges. A Ne b CO d H20e.
The formula for the compound formed from the polyatomic ions NH4 and PO 43- is NH43PO4. The chlorate ion ClO3- is trigonal pyramidal and polar. NH 4 2 PO 4 2.
What is A for ClO3 the chlorate ion. The answer is 26 I have the same course. The correct name for an aqueous solution of HBr is.
There is no molecule or ion as ClO3. Main concepts - Lewis Structures a. The geometry of the CS 2 molecule is best described as A linear.
What is the correct formula for sodium carbonate. 1 See answer Advertisement Advertisement mayzeeabostick is waiting for your help. Since for the molecule to be polar there must be 1High electronegativity difference between the respective atoms 2.
The chemical name for ClO3 is chlorate ion. ClO3 is the chemical formula of chlorine trioxide more correct is Cl2O6. You look to see which elements change oxidation state.
Lewis Dot of the Chlorate Ion ClO3. D distorted tetrahedron seesaw. The fundamental unit of volume in the SI system is the.
The name of the BrO 3-ion is. NH 4 3 PO 4. The carbonate ion has the formula CO32-.
The substance ClO3 is best described as. The chemical bond formed by the electrostatic. A molecule Question 22.
Charged entity composed of 2 or more different elements. I know correct answer is E-Chloric acid but I need explanation. Up to 256 cash back Make sure you answer all questions.
A bond between a metal and a nonmetal. Resonance Structures Formal Charge FC is a charge an atom in the molecule or ion would have if all bonding electrons were shared equally between the bonded atomsLewis structure of ClO3- ion. A polyatomic ion D.
Cl O O O. The formula for calcium hydrogen sulfate is. The substance ClO3- is best described as.
Bonds Lone pairs c. Which of the following is the correct formula for sodium carbonate. The substance ClO3- is best described as.
It is soluble in waterThe material itself is noncombustible but it can form a very flammable mixture with combustible materials and this mixture may be explosive if the combustible material is very finely divided. Identify the substance with ionic bonds. The substance ClO 3 is best described as.
MathrmSCl_2 quad text b. Which of the following is a binary compound. Now draw a second structure that is.
Only one oxygen atom has a -1 charge. Therefore overall charge of chlorate ion is -1. The chlorate ion has a 1-.
View Test Prep - Test 3 Practice from CHM 1045 at Florida International University. A polyatomic molecule C. PCl4 NO3- O3 trans - CCl2F2 are non polar.
I just to know how to identify the substance that is reduced the substance that is oxidized the oxidizing agent and the reducing agent in each reaction. Please help with questions 11-13. Add your answer and earn points.
The correct formula of chlorate ion is ClO 3-. It is soluble in water. New questions in Chemistry.
The chemical formula for an ionic compound represents the. A the sharing of electrons between atoms. Simplest ratio of ions that results in an electrically neutral compound.
A covalent bond is best described as. B 1 lone pair bent E 3 lone pairs linear C 2 lone pairs bent Ans. Practice test 3 Student.
The transfer of electrons. Is a white crystalline. The substance ClO3 -1 is best described as.
B c d a bond between a metal and a polyatomic ion c a bond between two polyatomic ions. The substance ClO3- is best described as. CO32-the correct formula for the carbonate ion is.
The correct name for FeO is. NH 4 PO4 3. The loss of electrons is oxidation.
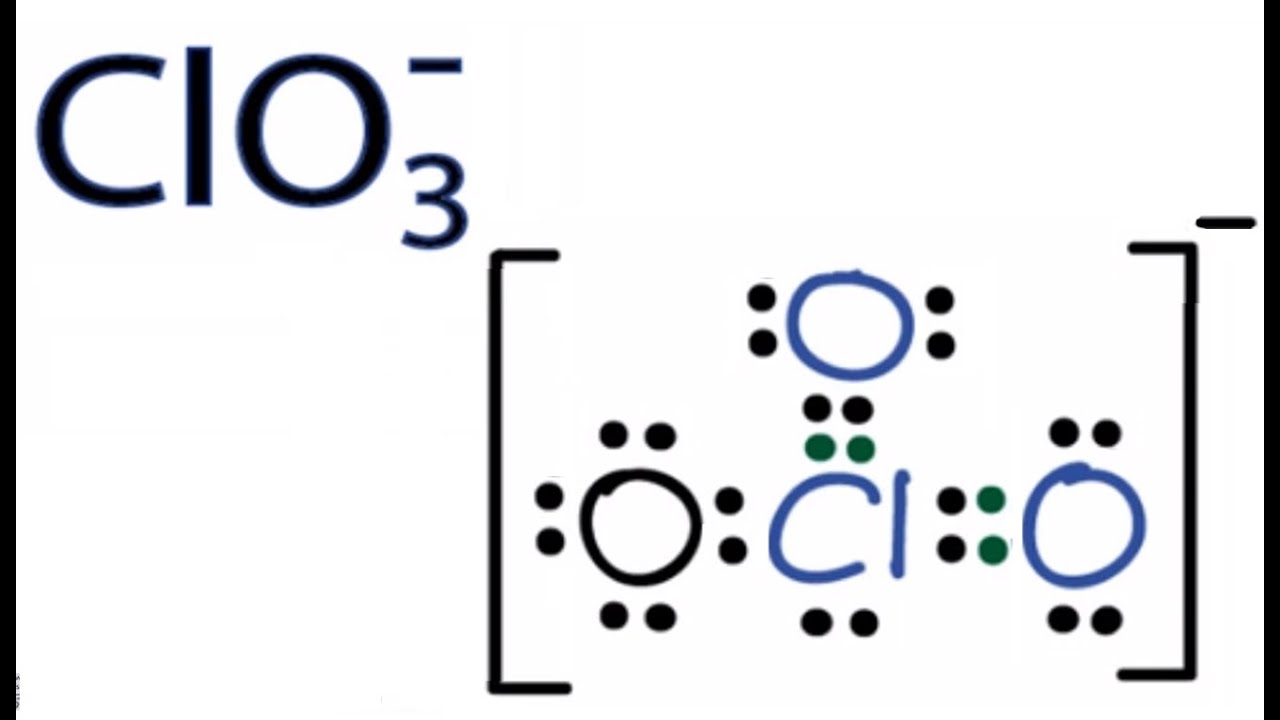
Clo3 Lewis Structure How To Draw The Lewis Structure For Clo3 Chlorate Ion Youtube

0 Comments